Application for Approval for Field Experiments Involving Modified Living Organisms (LMOs)
All field experiments involving modified living organisms (LMO) for research and development purposes must obtain a Certificate of Approval from the National Biosafety Board prior to commencing activities. Field experiments are a discharge activity because they involve the deliberate introduction of LMOs into the environment.
Approval Process
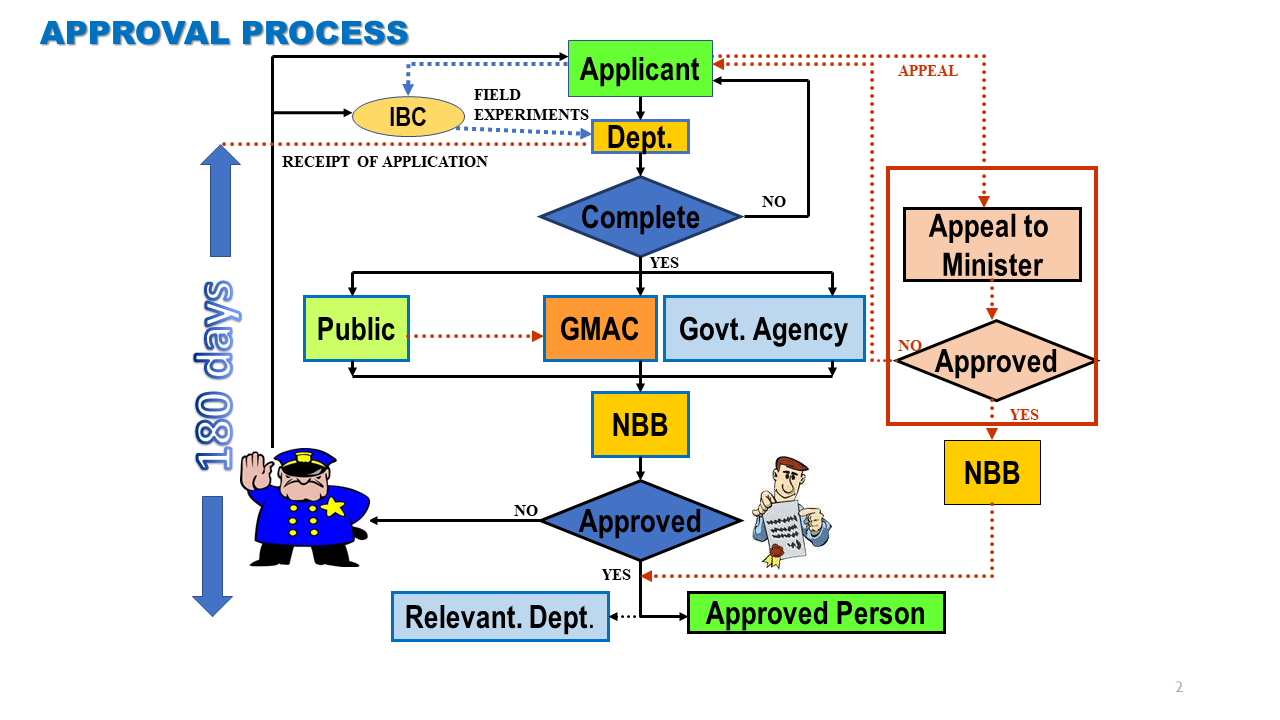
Fi
The fee charged depends on the size of the field experiment site
| FIELD TEST SITE SIZE | FI (RM) |
| Less than 5 ha | 100 |
| 5 ha - 10 ha | 250 |
| More than 10 ha | 500 |
The payment method is through remittance or bank draft under the name Secretary General, Ministry of Natural Resources and Environmental Sustainability and sent along with the application form.
Decision of the National Biosafety Board (NBB)
More information on the results can be obtained here.
Approval Application Form
a) Form A (Approval for Release Activities of LMO (Research and Development Purposes in All Field Experiments) or Importation of LMO that is Higher Plant)
Description:
This form is used to conduct field experiments involving modified living organisms (LMOs) consisting of high -grade plants. The LMO of the plant can be imported or produced locally for use as field testing material.
b) Form B (Approval for Release Activities of LMO (Research and Development Purposes in All Field Experiments) or Importation of LMO other than Higher Plants)
Description:
This form is used to conduct field experiments involving modified living organisms (LMOs) that are not composed of high -grade plants. The LMO can be imported or produced locally for use as field testing material.
c) Annex 2 (IBC Assessment of Project Proposal Involving Modern Biotechnology Activities)
Description:
This form should be used when the IBC evaluates the field test activities before submitting the approval application form to the Department of Biosafety.
d) LMO Controlled Field Experiment Guidelines
Description:
Provide researchers with procedures for conducting controlled field experiments for plants modified to comply with biosafety regulatory requirements.
e) Guidelines for the Environmental Risk Assessment of GM Plants in Malaysia
Description:
Provide guidance on the risk assessment of modified plants (GM) to the environment (ERA) in Malaysia. It covers ERA applications for the cultivation of GM plants, as well as the importation of food and animal nutrition using GM plants or products derived from GM plants.





